Brinzolamide 10 Mg/Ml Eye Drops Suspension

Package leaflet: Information for the patient
Brinzolamide 10 mg/ml Eye Drops, Suspension
Read all of this leaflet carefully before
you start using this medicine because it
contains important information for you.
• I t. You may need to
read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you only. Do not pass it on to others.
It may harm them, even if their signs of illness are the same as yours.
• If you get any sideeffects, talk to your doctor or pharmacist. This includes any possible side effects not listed in thi t. See section 4.
Wha
1. What Brinzolamide is and what it is used for
2. What you need to know before you use Brinzolamide
3. How to use Brinzolamide
4. Possible side effects
5. How to store Brinzolamide
6. Contents of the pack and other information
1. What Brinzolamide is and what it is used for
Brinzolamide contains the active ingredient brinzolamide which belongs to a group of medicines called carbonic anhydrase inhibitors. It reduces pressure within the eye.
Brinzolamide is used to treat high pressure in the eye. This pressure can lead to an illness called glaucoma.
If the pressure in the eye is too high, it can damage your sight.
2. What you need to know before you use Brinzolamide
Do not use Brinzolamide:
• if you have severe kidney problems
• if you are allergic to brinzolamide or any of the other ingredients of this medicine (listed in section 6)
• if you are allergic to medicines called sulfonamides. Examples include medicines used to treat diabetes and infections and also diuretics (water tablets). Brinzolamide may cause the same allergy.
• if you have too much acidity in your blood (a condition called hyperchloraemic acidosis).
If you have further questions ask your doctor for advice.
Warnings and precautions
Talk to your doctor or pharmacist or nurse
before using Brinzolamide:
• if you have kidney or liver problems
• if you have dry eyes or cornea problems
• if you are taking other sulphonamide medicines
• if you have been told you have the following eye conditions: pseudoexfoliative, pigmentary or narrow angle glaucoma.
Children and adolescents
Brinzolamide is not to be used by infants,
children or adolescents under 18 years of
age unless advised by your doctor.
Other medicines and Brinzolamide
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines including medicines obtained without a prescription.
• if you are taking another carbonic anhydrase inhibitor (acetazolamide or dorzolamide), talk to your doctor.
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine.
You should not use Brinzolamide if you are pregnant, or might become pregnant.
Your doctor will advise if you can use brinzolamide while you are breast-feeding as it is not known whether it can be excreted in milk.
Driving and using machines
You ma nd that your vision is blurred or have other changes to your vision for a time just after you use Brinzolamide. Do not drive or use machines until your vision is clear.
Brinzolamide may impair the ability of patients to perform tasks requiring mental alertness and/or physical coordination. If affected, take care when driving or using machines.
Brinzolamide contains benzalkonium chloride
Benzalkonium chloride may cause eye irritation and is known to discolour soft contact lenses. Avoid contact with soft contact lenses. Remove contact lenses prior to application of Brinzolamide and wait at least 15 minutes after applying the dose before reinsertion.
3. How to use Brinzolamide
Always use this medicine exactly as your doctor or pharmacist has told you. Check with your doctor or pharmacist if you are not sure.
The recommended dose is 1 drop in the affected eye or eyes, twice a day -morning and night.
Use this much unless your doctor told you t erent. Only use
Brinzolamide in both eyes if your doctor told you to. Use it for as long as your doctor told you to.
Only use this medicine for dropping in your eyes - do not swallow.
See below for more advice
1
2
3
1. Get the eye drops bottle. You must not use the bottle if the tamper-proof seal on the bottle neck is broken before
y e.
2. Wash your hands.
3. Shake the bottle well and twist off the cap.
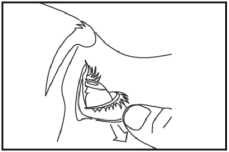
4. Bend your head backwards and gently pull your lower eyelid down until there is a small "pocket" (picture 1). It is easier if you sit or stand in front of a mirror.
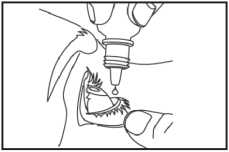
5. Hold the bottle upside down between your thumb and forefinger, above one eye. Squeeze one drop into the formed pocket. Do not touch your eye or eyelashes, surrounding areas or anything else with the dropper tip (picture 2).
6. If a drop misses your eye, try again.
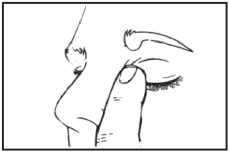
7. Let go of the eyelid and gently press
to the corner of your eye, by the nose. This helps to stop Brinzolamide getting into the rest of the body (picture 3).
8. If you take drops in both eyes, repeat the steps 4 to 7 for your other eye.
9. Tighten the cap on the bottle immediately after use.
10. Use up one bottle before opening the next bottle.
If you are using other eye drops or ointment,
between putting in Brinzolamide and the other drops. If you use an eye ointment, this should always be applied last.
If you use more Brinzolamide than you should
If you use more Brinzolamide than you should, rinse it all out with warm water.
Do not put in any more drops until it is time for your next regular dose.
If you forget to use Brinzolamide
If you forget to use Brinzolamide, use a single drop as soon as you remember, and then go back to your regular routine. Do not use a double dose to make up for a forgotten dose.
If you stop using Brinzolamide
If you stop using Brinzolamide without speaking to your doctor, the pressure in your eye will not be controlled which could lead to loss of sight.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everyone gets them. You can usually carry on using the drops unless the effects are serious.
513511



Proof No

|
1 |
Main Font |
Myriad Pro |
Body Text Size |
9 pt | |
|
1_ |
Dimensions |
140 X 460 mm | |||
Stop using Brinzolamide immediately if you get any of the following effects and go straight away to hospital or seek medical advice from your doctor:
Common: may affect up to 1 in 10 people:
• eye pain, redness of the eye, blurred vision, increased tear production with possible sensitivity to light, or a feeling that something is in the eye.
Uncommon: may affect up to 1 in 100 people:
• < ty breathing, increase in rate of breathing with sweating, wheezing or crackling sounds. You may also have blue lips, pale or grey skin. This may indicate you are not getting enough oxygen into the body.
Rare: may affect up to 1 in 1,000 people:
• heavy or pressing sensation on your chest with chest pain. This could be signs of problems with your heart such as angina.
Not known: cannot be estimated from the available data:
• allergic reactions that may appear as
a rash, swelling of the eye, eyelid, lips, tongue, face, hands or throat which ma ty breathing.
Other side effects include
Common: may affect up to 1 in 10 people:
• bitter or unusual taste in your mouth
• eye irritation.
Uncommon: may affect up to 1 in 100 people:
• redness ky skin and inflammation on eyelids
• eye sensitivity to light difficulty seeing in the presence of very bright light
• < ation or infection of the eye
• dry or itchy eye
• eye discharge
• < ation of the eyelid glands
• abnormal eye sensation
• growth on surface of eye
• increased pigmentation of the eye or a grey coloured band around the eye
• fatty or mineral deposits in or around the eye
• tired eyes
• eyelid crusting
• increased tear production
• decreased heart rate, palpitations, which can be felt as a thumping in your chest
• shortness of breath or a tightness of the chest with wheezing sometimes brought on by exercise
• decreased red blood cell count in the blood. You may experience tiredness, shortness of breath, cold hands and feet, pale skin and difficulty in healing after a cut
• blood tests show an increased chloride level in the blood
• dizziness
• depression or lack of enthusiasm or interest
• tingling or pins and needles sensations in the hands or feet
• having nightmares
• nervousness
• fatigue
• decreased sex drive or problems getting or maintaining an erection
• cold symptoms
• < ty breathing through the nose with pain in the face (these may be signs of sinus problems)
• throat irritation, pain or sore throat
• mucus build up in back of nose or throat
• abnormal or decreased sensation in mouth
• inflammation of the lining of the food pipe with pain
• abdominal pain, nausea, vomiting, upset stomach or indigestion
• frequent bowel movements, diarrhoea or intestinal gas
• digestive disorder
• kidney pain
• headache
• muscle pain or muscle spasms
• back pain
• < ty walking or lifting
• nose bleeds, runny nose, or sneezing
• red rash or an red area of skin with small bumps
• skin tightness
• < ye
• dry mouth.
Rare: may affect up to 1 in 1,000 people:
• < ty sleeping
• drowsiness
• increased pressure in eye
• damage to the optic nerve
• abnormal, double or reduced vision
• seeing flashing lights with discomfort or pain
• decreased eye sensation
• puffy eyes or swelling around the eyes
• irregular heart rate
• dr
• chest congestion or cough
• itchy rash (known as nettle rash) or hives
• itching
• loss of hair
• generalised weakness
• feeling jittery or irritable
• chest pain
• < ty with memory
• ringing in ears.
Not known: cannot be estimated from the available data:
• eyelid abnormality or redness around the eye
• decreased growth or number of eyelashes
• spinning sensation with a loss of balance (known as vertigo)
• increased or decreased blood pressure, increased heart rate
• abnormal liver blood tests
• frequent urination
• swelling of the hands or feet
• decreased sensation (can be also known as numbness)
• decreased taste sensation
• joint pain or pain in hands or feet
• shaking
• general feeling of being unwell
• decreased appetite.
Repor ects
If you get anyside effects, talk to your doctor or pharmacist or nurse. This includes any possible side effects not list t. You can also report
side effects directly via the Yellow Card Scheme at www.mhra.gov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Brinzolamide
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the bottle and box after "EXP". The expiry date refers to the last day of that month.
Keep the bottle in the outer carton.
You must throw away a bottle four weeks after y t, to
prevent infections. Write down the date you opened each bottle in the space below and on the bottle label and box.
For a pack containing a single bottle, write only one date.
Date opened (1):
Date opened (2):
Date opened (3):
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Brinzolamide contains
The active substance is brinzolamide. One ml of the eye drop suspension contains 10 mg of brinzolamide.
The other ingredients are: benzalkonium chloride, carbomer 974P, edetate disodium, mannitol (E421), purified water, sodium chloride, hydrochloric acid or sodium hydroxide for pH adjustment.
What Brinzolamide looks like and contents of the pack
Brinzolamide Eye Drops, Suspension is a white t te liquid.
Brinzolamide is available in a 5 ml plastic
(low density polyethylene) bottle, containing 5 ml of Brinzolamide Eye Drops, Suspension, with a plastic insert dropper and a plastic cap.
Brinzolamide is available in the following pack sizes:
1 x 5 ml bottle packed in a single carton 3 x 5 ml bottles packed in a single carton
Not all pack sizes may be marketed.
Marketing Authorisation Holder
Mylan, Potters Bar, Hertfordshire, EN6 1TL,
United Kingdom
Manufacturer
Lusomedicamenta Sociedade Tecnica Farmaceutica, S.A., Rua Norberto de Oliveira, no 1/5, Povoa de Santo Adriao, 2620-111, Portugal
This leaflet was last revised in 03/2015
513511


Proof No

|
1 |
Main Font |
Myriad Pro |
Body Text Size |
9 pt | |
|
1_ |
Dimensions |
140 X 460 mm | |||