Cetirizine Dihydrochloride 10 Mg Film-Coated Tablets
SUMMARY OF PRODUCT CHARACTERISTICS
1 NAME OF THE MEDICINAL PRODUCT
Cetirizine dihydrochloride 10 mg film-coated tablets
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
One film-coated tablet contains 10 mg cetirizine dihydrochloride.
Excipients: one film-coated tablet contains 100.20 mg lactose-monohydrate. For a full list of excipients, see section 6.1.
3 PHARMACEUTICAL FORM
Film-coated tablets.
White coloured, circular, biconvex film coated tablet. Marked with ‘A’ on one side and a break-line on the other.
4 CLINICAL PARTICULARS
4.1 Therapeutic indications
Adults and adolescents over 12 years of age:
Symptomatic treatment of allergic rhinitis (seasonal and perennial) associated allergic conjunctivitis, and chronic idiopathic urticaria.
Children 6-12 years:
Symptomatic treatment of allergic rhinitis (seasonal and perennial), and chronic idiopathic urticaria.
4.2
Posology and method of administration
Posology
Paediatric population Children aged from 6 to12 years:
1 tablet (10 mg) once daily or / tablet (5mg) taken twice daily (morning and evening)
For children weighing less than 30 kg:
/ tablet (5mg) taken once daily.
Clinical trials in children have not exceeded four weeks.
Adults and adolescents over 12 years of age: 1 tablet (10 mg) once daily
If drowsiness occurs, the tablet can be administered in the evening.
Elderly subjects: data do not suggest that the dose needs to be reduced in elderly subjects provided that the renal function is normal. The duration of the treatment may vary depending on the symptoms.
Cetirizine is contraindicated in patients with severe renal impairment.
Patients with moderate to severe renal impairment: there are no data to document the efficacy/safety ratio in patients with renal impairment. Since cetirizine is mainly excreted via renal route (see section 5.2), in cases no alternative treatment can be used, the dosing intervals must be individualized according to renal function. Refer to the following table and adjust the dose as indicated. To use this dosing table, an estimate of the patient’s creatinine clearance (CLcr) in ml/min is needed. The CLcr (ml/min) may be estimated from serum creatinine (mg/dl) determination using the following formula:
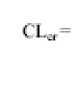
[] 40 - age{ i>ears} ] x weight i fcg :■. ^ 7 2 x serum t yen tinin e i mg ill i
0.S7 for women i
Dosing adjustments for adult patients with impaired renal function
|
Group |
Creatinine clearance (ml/min) |
Dosage and frequency |
|
Normal |
>80 |
10 mg once daily |
|
Mild |
50 - 79 |
10 mg once daily |
|
Moderate |
30 - 49 |
5 mg once daily |
|
Severe |
<30 |
5 mg once every 2 days |
|
End-stage renal disease -Patients undergoing dialysis |
<10 |
Contra-indicated |
In pediatric patients suffering from renal impairment, the dose will have to be adjusted on an individual basis taking into account the renal clearance of the patient, his age and his body weight.
Patients with hepatic impairment: no dose adjustment is needed in patients with solely hepatic impairment.
Patients with hepatic impairment and renal impairment: dose adjustment is recommended (see Patients with moderate to severe renal impairment above).
Method of administration For oral use.
4.3 Contraindications
Cetirizine hydrochloride 10 mg Film Coated Tablets are contraindicated in:
- Patients with a history of hypersensitivity to any of the constituents of the formulation, to hydroxyzine or to any piperazine derivatives.
- Patients with severe renal impairment at less than 10mL/min creatinine clearance.
4.4 Special warnings and precautions for use
In some patients, long term treatment with cetirizine tablets may lead to an increased risk of caries due to mouth dryness. The patients should therefore be informed about the importance of oral hygiene.
At impaired hepatic function and renal function, the elimination of cetirizine may be impaired. Caution should be exercised when administering cetirizine to these patients. (see section 4.2 posology and section 4.3 contraindications).
At therapeutic doses, no clinically significant interactions have been demonstrated with alcohol (for a blood alcohol level of 0.5 g/L). Nevertheless, precaution is recommended if alcohol is taken concomitantly.
Caution should be taken in patients with predisposition factors of urinary retention (e.g. spinal cord lesion, prostatic hyperplasia) as cetirizine may increase the risk of urinary retention.
Caution is recommended in epileptic patients and patients at risk of convulsions.
The use of the film-coated tablet formulation is not recommended in children aged less than 6 years since this formulation does not allow for appropriate dose adaptation.
Allergy skin tests are inhibited by antihistamines and a wash-out period (of 3 days) is required before performing them.
Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency of glucose-galactose malabsorption should not take cetirizine film-coated tablets.
The use of the film-coated tablet formulation is not recommended in children aged less than 6 years since this formulation does not allow for appropriate dose adaptation.
4.5 Interaction with other medicinal products and other forms of interaction
Due to the pharmacokinetic, pharmacodynamic and tolerance profile of cetirizine, no interactions are expected with this antihistamine. Actually, neither pharmacodynamic nor significant pharmacokinetic interaction was reported in drug-drug interactions studies performed, notably with pseudoephedrine or theophylline (400 mg/day).
The extent of absorption of cetirizine is not reduced with food, although the rate of absorption is decreased.
Caution is recommended with the concomitant use of CNS depressants.
4.6 Fertility, pregnancy and lactation
Pregnancy
For cetirizine very rare clinical data on exposed pregnancies are available. The data indicates no adverse effects of cetirizine on pregnancy or on the health of the foetus/new born child. To date no other relevant epidemiological data are available.
Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryonal/fetal development, parturition or postnatal development. Caution should be exercised when prescribing to pregnant women.
Breast-feeding
No data concerning the excretion of cetirizine into human milk are available. Cetirizine is excreted in human milk at concentrations representing 0.25 to 0.90 those measured in plasma, depending on sampling time after administration. Therefore, caution should be exercised when prescribing cetirizine to lactating women.
4.7 Effects on ability to drive and use machines
Objective measurements of driving ability, sleep latency and assembly line performance have not demonstrated any clinically relevant effects at the recommended dose of 10 mg.
Patients intending to drive, engaging in potentially hazardous activities or operating machinery should not exceed the recommended dose and should take their response to the medicinal product into account. In these sensitive patients, concurrent use with alcohol or other CNS depressants may cause additional reductions in alertness and impairment of performance.
4.8 Undesirable effects
Clinical studies have shown that cetirizine at the recommended dosage has minor undesirable effects on the CNS, including somnolence, fatigue, dizziness and headache. In some cases, paradoxical CNS stimulation has been reported.
Although cetirizine is a selective antagonist of peripheral H1-receptors and is relatively free of anticholinergic activity, isolated cases of micturition difficulty, eye accommodation disorders and dry mouth have been reported.
Instances of abnormal hepatic function with elevated hepatic enzymes accompanied by elevated bilirubin have been reported. Mostly this resolves upon discontinuation of the treatment with cetirizine dihydrochloride.
Clinical trials
Double blind controlled clinical or pharmacoclinical trials comparing cetirizine to placebo or other antihistamines at the recommended dosage (10 mg daily for cetirizine), of which quantified safety data are available, included more than 3200 subjects exposed to cetirizine.
From this pooling, the following adverse events were reported for cetirizine 10 mg in the placebo-controlled trials at rates of 1.0 % or greater:
|
Adverse event |
Cetirizine 10 |
Placebo |
|
(WHO-ART) |
mg (n= 3260) |
(n = 3061) |
|
Body as a whole - general disorders Fatigue |
1.63 % |
0.95 % |
|
Central and peripheral nervous system disorders |
1.10 % |
0.98 % |
|
Dizziness Headache |
7.42 % |
8.07 % |
|
Gastro-intestinal system disorders Abdominal pain |
0.98 % |
1.08 % |
|
Dry mouth |
2.09 % |
0.82 % |
|
Nausea |
1.07 % |
1.14 % |
|
Psychiatric disorders Somnolence |
9.63 % |
5.00 % |
|
Respiratory system disorders Pharyngitis |
1.29 % |
1.34 % |
Although statistically more common than under placebo, somnolence was mild to moderate in the majority of cases. Objective tests as demonstrated by other studies have demonstrated that usual daily activities are unaffected at the recommended daily dose in healthy young volunteers.
Adverse drug reactions at rates of 1 % or greater in children aged from 6 months to 12 years, included in placebo-controlled clinical or pharmacoclinical trials are:
|
Adverse drug reactions (WHO-ART) |
Cetirizine (n=1656) |
Placebo (n =1294) |
|
Gastro-intestinal system disorders |
|
Diarrhoea |
1.0 % |
0.6 % |
|
Psychiatric disorders Somnolence |
1.8 % |
1. 4 % |
|
Respiratory system disorders Rhinitis |
1.4 % |
1.1 % |
|
Body as a whole - general disorders Fatigue |
1.0 % |
0.3 % |
Post-marketing experience
In addition to the adverse effects reported during clinical studies and listed above, isolated cases of the following adverse drug reactions have been reported in postmarketing experience.
Undesirable effects are described according to MedDRA System Organ Class and by estimated frequency based on post-marketing experience.
Frequencies are defined as follows: Very common (>1/10); common (> 1/100 to <1/10); uncommon (>1/1,000 to <1/100); rare (>1/10,000 to <1/1,000); very rare (<1/10,000), not known (cannot be estimated from the available data)
Blood and lymphatic disorders:
Very rare: thrombocytopenia
Immune system disorders:
Rare: hypersensitivity, allergic reactions (see Skin and subcutaneous disorders)
Very rare: anaphylactic shock
Metabolism and nutrition disorders:
Not known: increased appetite
Psychiatric disorders:
Uncommon: agitation
Rare: aggression, confusion, depression, hallucination, insomnia Very rare: tics, suicidal ideation.
Nervous system disorders:
Uncommon: paraesthesia Rare: convulsions
Very rare: dysgeusia, syncope, tremor, dystonia, dyskinesia Not known: amnesia, memory impairment
Eye disorders:
Rare: abnormal involuntary eye movements.
Very rare: accommodation disorder, blurred vision, oculogyration
Ear and labyrinth disorders:
Not known: vertigo
Cardiac disorders:
Rare: tachycardia
Gastro-intestinal disorders:
Uncommon: diarrhoea
Hepatobiliary disorders:
Rare: abnormal hepatic function (increased transaminases, alkaline phosphatase, y-GT and bilirubin)
Skin and subcutaneous tissue disorders:
Uncommon: skin rash, pruritus,
Rare: urticaria
Very rare: fixed drug eruption, erythema multiforme, angioneurotic oedema,
Renal and urinary disorders:
Very rare: dysuria, enuresis, micturition difficulties Not known: urinary retention
General disorders and administration site conditions:
Uncommon: asthenia, malaise Rare: oedema
Investigations:
Rare: weight increased
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard.
4.9 Overdose
There is limited experience of overdosing, 20 mg to a 2-year-old, 30 mg to a 3-year-old and 40 mg to an 11-year-old did not give any symptoms. 60 mg to a 4-year-old gave mild intoxication, 400 mg to a 14-year-old gave mild symptoms, while 400 to 500 mg to an adult gave no symptoms at all
Symptoms
Symptoms observed after an overdose of cetirizine are mainly associated with CNS effects or with effects that could suggest an anticholinergic effect.
Adverse events reported after an intake of at least 5 times the recommended daily dose are: confusion, diarrhoea, dizziness, fatigue, headache, malaise, mydriasis, pruritus, restlessness, sedation, somnolence, stupor, tachycardia, tremor, and urinary retention.
Management
There is no known specific antidote to cetirizine.
Should overdose occur, symptomatic or supportive treatment is recommended.
The patient should be kept under clinical observation for at least four hours after ingestion, and the blood pressure, heart rate and vital signs monitored until stable. In symptomatic cases, ECG should be performed.
Gastric lavage should be considered following ingestion of a short occurrence.
Oral activated charcoal (50 g for an adult, 10-15 g for a child) should be considered if more than 2.5 mg/kg cetirizine has been ingested within one hour
Cetirizine is not effectively removed by dialysis.
5.1 Pharmacodynamic properties
ATC code: R06A E07 Mechanism of action
Pharmacotherapeutic group: Antihistamine for systemic use, piperazine derivative Cetirizine hydrochloride is a racemate and an anti-allergic with specific histamine H1-receptor blocking characteristics.
Cetirizine, a human metabolite of hydroxyzine, is a potent and selective antagonist of peripheral H1-receptros. In vitro receptor binding studies have shown no measurable affinity for other than H1-receptors.
Pharmacodynamic effects
Cetirizine inhibits cutaneous reactions in allergic individuals by VIP (Vasoactive Intestinal Polypeptide) and the P substance, neuropeptides that are considered involved in the allergic reaction. Effect is reached within 2 hours with a maximum effect after 4 hours, and remains for at least 24 hours. In allergic individuals, at a dose of 10mg once or twice daily, cetirizine inhibits the late phase recruitment of eosinophils, in the skin and conjunctiva of atopic subjects after simulation with allergens and unselective histamine liberators, by a mechanism that is not primarily explained by the H1-receptor blocking characteristics of the pharmaceutical.
Clinical efficacy and safety
Studies in healthy volunteers show that cetirizine, at doses of 5 and 10mg strongly inhibits the wheal and flare reactions induced by very high concentrations of histamine into the skin, but the correlation with efficacy is not established.
Paediatric population
In a 35-day study in children aged 5 to 12, no tolerance to the antihistaminic effect (suppression of wheal and flare) of cetirizine was found. When a treatment with cetirizine is topped after repeated administration, the skin recovers its normal reactivity to histamine within 3 days.
In a six-week, placebo-controlled study of 186 patients with allergic rhinitis and concomitant mild to moderate asthma, cetirizine 10mg once daily improved rhinitis symptoms and did not alter pulmonary function. This study supports the safety of administering cetirizine to allergic patients with mild to moderate asthma.
In a placebo-controlled study, cetirizine given at the high daily dose of 60mg for seven days did not cause statistically significant prolongation of QT interval.
At the recommended dosage, cetirizine has demonstrated that it improves the quality of life of patients with perennial and seasonal allergic rhinitis.
5.2 Pharmacokinetic properties
Absorption
Cetirizine is absorbed with small inter-individual variations.
Distribution
Cetirizine has not been given intravenously, therefore the bioavailability, clearance and distribution volume (Vd) are unknown. Maximum plasma concentration is achieved within 1 hour and the terminal half-life is about 10 hours in adults and 6 hours in children between ages of 6-12 years. The grade of protein binding in plasma is about 93%.
Biotransformation
Cetirizine is metabolised to a small extent with a known inactive main metabolite. Elimination
Cetirizine is eliminated to 60% in unchanged form via the kidneys within 96 hours. At repeated administration there is no accumulation at hand, nor is absorption or elimination affected. With impaired kidney function, the elimination is slower and the half-life is prolonged. Elimination will also be decreased in cases of hepatic impairment. There is no evidence that the pharmacokinetics of cetirizine is altered in elderly patients unless renal or hepatic function is reduced.
Linearity
Cetirizine exhibits linear kinetics over the range of 5 to 60mg.
Special populations Elderly:
Following a single 10mg oral dose, half-life increased by about 50% and clearance decreased by 40% in 16 elderly subjects compared to the normal subjects. The decrease in cetirizine clearance in these elderly volunteers appeared to be related to their decreased renal function.
Paediatric Population:
The half-life of cetirizine was about 6 hours in children of 6-12 years and 5 hours in children 2-6 years. In infants and toddlers aged 6 to 24 months, it is reduced to 3.1 hours.
Renal Impairment:
The pharmacokinetics of the drug were similar in patients with mild impairment (creatinine clearance higher than 40 mL/min) and healthy volunteers. Patients with moderate renal impairment had a 3-fold increase in half-life and 70% decrease in clearance compared to healthy volunteers.
Patients on hemodialysis (creatinine clearance less than 7 mL/min) given a single oral 10mg dose of cetirizine had a 3-fold increase in half-life and a 70% decrease in clearance compared to normal. Cetirizine was poorly cleared by haemodialysis. Dosing adjustment is necessary in patients with moderate or severe renal impairment (see section 4.2).
Hepatic Impairment:
Patients with chronic liver diseases (hepatocellular, cholestatic, and biliary cirrhosis) given 10 or 20mg of cetirizine as a single dose had a 50% increase in half-life along with a 40% decrease in clearance compared to healthy subjects.
Dosing adjustment is only necessary in hepatically impaired patients if concomitant renal impairment is present.
5.3 Preclinical safety data
Preclinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, toxicity to reproduction, genotoxicity or carcinogenicity.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Tablet core
Lactose monohydrate
Microcrystalline cellulose
Colloidal anhydrous silica
Maize starch
Talc
Magnesium stearate Coating
Titanium dioxide (E171)
Hypromellose Lactose monohydrate Macrogol Sodium citrate
6.2 Incompatibilities
Not applicable
6.3 Shelf life
3 years
6.4 Special precautions for storage
This medicinal product does not require any special storage conditions.
6.5. Nature and contents of container
Blister comprising of PVC/Aluminium foil with 7, 14 and 30 tablets. Not all packs may be marketed.
6.6 Special precautions for disposal
No special requirements.
7 MARKETING AUTHORISATION HOLDER
Fannin (UK) Limited 42-46 Booth Drive Park Farm South Wellingborough Northamptonshire NN8 6GT UK
8 MARKETING AUTHORISATION NUMBER(S)
PL 20417/0020
9 DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
13/10/2009
10 DATE OF REVISION OF THE TEXT
08/03/2016