Levetiracetam Beacon 100 Mg/Ml Oral Solution
|
Weight |
Starting dose: 0.1 ml/kg twice daily |
Maximum dose: 0.3 ml/kg twice daily |
|
6 kg |
0.6 ml twice daily |
1.8 ml twice daily |
|
8 kg |
0.8 ml twice daily |
2.4 ml twice daily |
|
10 kg |
1 ml twice daily |
3 ml twice daily |
|
15 kg |
1.5 ml twice daily |
4.5 ml twice daily |
|
20 kg |
2 ml twice daily |
6 ml twice daily |
|
25 kg |
2.5 ml twice daily |
7.5 ml twice daily |
|
From 50 kg |
5 ml twice daily |
15 ml twice daily |
|
Weight |
Starting dose: 0.07 ml/kg twice daily |
Maximum dose: 0.21 ml/kg twice daily |
|
4 kg |
0.3 ml twice daily |
0.85 ml twice daily |
|
5 kg |
0.35 ml twice daily |
1.05 ml twice daily |
|
6 kg |
0.45 ml twice daily |
1.25 ml twice daily |
|
7 kg |
0.5 ml twice daily |
1.5 ml twice daily |

Package Leaflet: Information for the patient
Levetiracetam 100 mg/ml oral solution
Levetiracetam
Read all of this leaflet carefully before you or your
child start taking this medicine because it contains
important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What Levetiracetam is and what it is used for
2. What you need to know before you take Levetiracetam
3. How to take Levetiracetam
4. Possible side effects
5. How to store Levetiracetam
6. Contents of the pack and other information
1. What Levetiracetam is and what it is used for
Levetiracetam is an antiepileptic medicine (a medicine
used to treat seizures in epilepsy).
Levetiracetam is used
• on its own in adults and adolescents from 16 years of age with newly diagnosed epilepsy, to treat a certain form of epilepsy. Epilepsy is a condition where the patients have repeated fits (seizures). Levetiracetam is used for the epilepsy form in which the fits initially affect only one side of the brain, but could thereafter extend to larger areas on both sides of the brain (partial onset seizure with or without secondary generalisation). Levetiracetam has been given to you by your doctor to reduce the number of fits.
• as an add-on treatment to other antiepileptic medicines to treat:
■ partial onset seizures with or without generalisation in adults, adolescents, children and infants from one month of age
■ myoclonic seizures (short, shock-like jerks of a muscle or group of muscles) in adults and adolesecents from 12 years of age with juvenile myoclonic epilepsy.
■ primary generalised tonic-clonic seizures (major fits, including loss of consciousness) in adults and adolescents from 12 years of age with idiopathic generalised epilepsy (the type of epilepsy that is thought to have a genetic cause).
2. What you need to know before you take Levetiracetam
Do not take Levetiracetam
• If you are allergic to levetiracetam, pyrrolidone derivatives or any of the other ingredients of this medicine (listed in Section 6).
Warnings and precautions
Talk to your doctor before taking Levetiracetam
• If you suffer from kidney problems, follow your doctor's instructions. He/she may decide if your dose should be adjusted.
• If you notice any slow down in the growth or unexpected puberty development of your child, please contact your doctor.
• A small number of people being treated with antiepileptics such as Levetiracetam have had thoughts of harming or killing themselves. If you have any symptoms of depression and/or suicidal ideation, please contact your doctor.
Children and adolescents
Levetiracetam is not indicated in children and adolescents below 16 years on its own (monotherapy)
Other medicines and Levetiracetam
Tell your doctor or pharmacist if you are taking or have recently taken or might take any other medicines. Do not take macrogol (a drug used as laxative) for one hour before and one hour after taking levetiracetam as this may result in a reduction of its effect.
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant, or are planning to have a baby, ask your doctor for advice before taking this medicine. Levetiracetam should not be used during pregnancy unless clearly necessary. A risk of birth defects for your unborn child cannot be completely excluded. Levetiracetam has shown unwanted reproductive effects in animal studies at dose levels higher than you would need to control your seizures.
Breast-feeding is not recommended during treatment.
Driving and using machines
Levetiracetam may impair your ability to drive or operate any tools or machinery, as it may make you feel sleepy. This is more likely at the beginning of treatment or after an increase in the dose. You should not drive or use machines until it is established that your ability to perform such activities is not affected.
Levetiracetam contains methyl parahydroxybenzoate and maltitol
Levetiracetam oral solution includes methyl parahydroxybenzoate (E218) which may cause allergic reactions (possibly delayed).
Levetiracetam oral solution also contains maltitol liquid (E965). If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicinal product.
3. How to take Levetiracetam
Always take this medicine exactly as your doctor or pharmacist has told you.
Check with your doctor or pharmacist if you are not sure.
Levetiracetam must be taken twice a day, once in the morning and once in the evening, at about the same time each day. Take the oral solution following your doctor's instructions.
Monotherapy
Dose in adults and adolescents (from 16 years of age):
General dose: between 10 ml (1,000 mg) and 30 ml (3,000 mg) each day, divided in 2 intakes per day. When you will first start taking Levetiracetam, your doctor will prescribe you a lower dose (500mg each day) during 2 weeks before giving you the lowest general dose of 1000mg.
Add-on therapy
Dose in adults and adolescents (12 to 17 years) weighing 50 kg or more:
General dose: between 10 ml (1,000 mg) and 30 ml (3,000 mg) each day divided in 2 intakes per day.
Dose in infants (6 to 23 months), children (2 to 11 years) and adolescents (12 to 17 years) weighing less than 50 kg:
Your doctor will prescribe the most appropriate pharmaceutical form of levetiracetam according to the age, weight and dose.
General dose: between 0.2 ml (20 mg) and 0.6 ml (60 mg) per kg bodyweight each day, divided in 2 intakes per day.
The exact quantity of oral solution formulation should be delivered using the syringe provided in the cardboard box.
Dose in infants (1 month to less than 6 months):
General dose: between 0.14 ml (14 mg) and 0.42 ml (42 mg) per kg bodyweight each day, divided in 2 intakes per day. The exact quantity of oral solution formulation should be delivered using the syringe provided in the cardboard box.
Method of Administration:
Levetiracetam may be diluted in a glass of water or baby's bottle. You may take Levetiracetam with or without food.
Instructions for use:
• Open the bottle: press the cap and turn it anticlockwise (figure 1)
• Separate the adaptor from the syringe (figure 2) Insert the adaptor into the bottle neck. Ensure it is well fixed.

• Take the syringe and put it in the adaptor opening (figure 2). Turn the bottle upside down (figure 3)
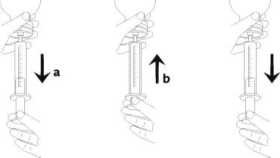
• Fill the syringe with a small amount of solution by pulling the piston down (figure 4 a), then push the piston upward in order to remove any possible bubble (figure 4 b) Pull the piston down to the graduation mark corresponding to the quantity in milliliters (ml) prescribed by your doctor (figure 4 c).
O
• Turn the bottle the right way up. Remove the syringe from the adaptor.
• Empty the contents of the syringe in a glass of water or baby's bottle by pushing the piston to the bottom of the syringe (figure 5).
• Drink the whole contents of the glass/baby's bottle.
• Close the bottle with the plastic screw cap.
• Wash the syringe with water (figure 6)

Duration of treatment:
• Levetiracetam is used as a chronic treatment. You should continue Levetiracetam treatment for as long as your doctor has told you.
• Do not stop your treatment without your doctor's advice as this could increase your seizures.
If you take more Levetiracetam than you should:
The possible side effects of an overdose of Levetiracetam are sleepiness, agitation, aggression, decrease of alertness, inhibition of breathing and coma. Contact your doctor if you took more than you should. Your doctor will establish the best possible treatment of overdose.
If you forget to take Levetiracetam:
Contact your doctor if you have missed one or more doses.
Do not take a double dose to make up for a forgotten dose.
If you stop taking Levetiracetam:
If stopping treatment, Levetiracetam should be discontinued gradually to avoid an increase of seizures. Should your doctor decide to stop your Levetiracetam treatment, he/she will instruct you about the gradual withdrawal of Levetiracetam.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
The most frequently reported adverse reactions were nasopharyngitis, somnolence (sleepiness), headache, fatigue and dizziness. At the beginning of the treatment or at dose increase side effects like sleepiness, tiredness and dizziness may be more common. These effects should however decrease over time.
Very common: may affect more than 1 user in 10 people
• nasopharyngitis;
• somnolence (sleepiness), headache.
Common: may affect 1 to 10 users in 100 people
• anorexia (loss of appetite);
• depression, hostility or aggression, anxiety, insomnia, nervousness or irritability;
• convulsion, balance disorder (equilibrium disorder), dizziness (sensation of unsteadiness), lethargy (lack of energy and enthusiasm), tremor (involuntary trembling);
• vertigo (sensation of rotation);
• cough;
• abdominal pain, diarrhoea, dyspepsia (indigestion), vomiting, nausea;
• rash;
• asthenia/fatigue (tiredness).
Uncommon: may affect 1 to 10 users in 1,000 people
• decreased number of blood platelets, decreased number of white blood cells;
• weight decrease, weight increase;
• suicide attempt and suicidal ideation, mental disorder, abnormal behaviour, hallucination, anger, confusion, panic attack, emotional instability/mood swings, agitation;
• amnesia (loss of memory), memory impairment (forgetfulness), abnormal coordination/ataxia (impaired coordinated movements), paraethesia (tingling), disturbance in attention (loss of concentration);
• diplopia (double vision), vision blurred;
• elevated/abnormal values in a liver function test;
• hair loss, eczema, pruritis;
• muscle weakness, myalgia (muscle pain);
• injury.
Rare: may affect 1 to 10 users in 10,000 people
• infection;decreased number of all blood cell types;
• severe allergic reactions (DRESS, anaphylactic reaction [severe and important allergic reaction], Quincke's oedema [swelling of the face, lips, tongue and throat]);
• decreased blood sodium concentration;
• suicide, personality disorders (behavioural problems), thinking abnormal (slow thinking, unable to concentrate);
• uncontrollable muscle spasms affecting the head, torso and limbs, difficulty in controlling movements, hyperkinesia (hyperactivity);
• pancreatitis;
• liver failure, hepatitis;
• skin rash, which may form blisters and looks like small targets (central dark spots surrounded by a paler area, with a dark ring around the edge) (erythema multiforme), a widespread rash with blisters and peeling skin, particularly around the mouth, nose, eyes and genitals (Stevens-Johnson syndrome), and a more severe form causing skin peeling in more than 30% of the body surface (toxic epidermal necrolysis).
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. Also you can help to make sure that medicines remain as safe as possible by reporting any unwanted side effects via the internet at www.mhra.gov.uk/yellowcard. Alternatively you can call Freephone 0808 100 3352 (available from 10 a.m. to 2 p.m. Mondays to Fridays) or fill in a paper form available from your local pharmacy.
5. How to store Levetiracetam
Keep this medicine out of sight and reach of children.
Do not use this medicine after the expiry date stated on the carton box and bottle after EXP:
The expiry date refers to the last day of the month.
Do not use after 4 months of first opening the bottle. Store in the original bottle in order to protect from light.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Levetiracetam contains:
The active substance is called Levetiracetam. Each ml contains 100 mg of levetiracetam.
The other ingredients are:
Sodium citrate (for pH adjustment), Citric acid monohydrate (for pH adjustment), Methyl parahydroxybenzoate (E218), Glycerol (E422), Acesulfame potassium (E950), Maltitol liquid (E965), Raspberry liquid and Purified water.
What Levetiracetam looks like and the contents of the pack
Levetiracetam 100mg/ml oral solution is a clear liquid.
The 300 ml glass bottle of Levetiracetam (for children aged 4 years and above, adolescents and adults) is packed in a cardboard box containing a 10ml oral syringe (graduated every 0.25 ml) and an adaptor for the syringe.
The 150 ml glass bottle of Levetiracetam (for infants and young children aged from 6 months to less than 4 years) is packed in a cardboard box containing a 3ml oral syringe (graduated every 0.1 ml) and an adaptor for the syringe.
The 150 ml glass bottle of Levetiracetam (for infants aged 1 month to less than 6 months) is packed in a cardboard box containing a 1ml oral syringe (graduated every 0.05 ml) and an adaptor for the syringe.
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation Holder: Beacon Pharmaceuticals Ltd., 85 High Street, Tunbridge Wells, TN1 1YG, UK
Manufacturer: Remedica Ltd, Limassol Industrial Estate, CY-3508- Limassol, Cyprus
This leaflet was last revised in June 2016
HARD TO READ? CALL 01892 600930 FOR HELP
REXP257BNA
% Beacon
PHARMACEUTICALS