Tobravisc 3.0 Mg/Ml Eye Drops Solution
I
Read all of this leaflet carefully before you start using this medicine
• Keep this leaflet. You may need to read it again.
• If you have further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others.
It may harm them, even if their symptoms are the same as yours.
• If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.

1. What TOBRAVISC is and what it is used for
2. Before you use TOBRAVISC
3. How to use TOBRAVISC
4. Possible side effects
5. How to store TOBRAVISC
6. Further information
TOBRAVISC belongs to a group of medicines It is used for the treatment of bacterial known as aminoglycoside antibiotics. infections of the surface of the eye and eyelids,
such as conjunctivitis and blepharitis.
Do not use TOBRAVISC...
• If you are allergic to tobramycin, or any of the other ingredients listed in section 6.
• In CHILDREN under the age of 1 year.
Ask your doctor for advice.
Only use TOBRAVISC for dropping in your eye(s).
Take special care...
• If you suffer from liver or kidney disease.
Tell your doctor if you are taking any other antibiotic medicines by mouth or by injection.
• As with any antibiotic, use of TOBRAVISC for a long time may lead to other infections. If your symptoms get worse or suddenly return tell your doctor.
• If you experience allergic reactions with TOBRAVISC, discontinue use and consult your doctor. Allergic reactions may vary from localized itching or skin redness tc severe allergic reactions (anaphylactic reaction) or serious skin reactions. These allergic reactions may occur with other topical or systemic antibiotics of the same family (aminoglycosides)
Pregnancy and breast-feeding
TOBRAVISC should be used during pregnancy only if clearly needed. If you are pregnant, you think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before using this medicine. TOBRAVISC is not recommended during breastfeeding.
Driving and using machines
You may find that your vision is blurred for a time just after you use TOBRAVISC. Do not drive or use any machines unless your vision is clear.
Using other medicines
Tell your doctor or pharmacist if you are
taking or have recently taken any other medicines, including medicines obtained without a prescription.
If you are using other eye drops, wait at least 5-10 minutes after putting in TOBRAVISC before using the other drops. Put all your eye drops in before using any eye ointment.
Important Information if you wear Contact Lenses
Wearing contact lenses is not advisable during the treatment of an eye infection, as they may make your condition worse.
Do not use the drops while wearing contact lenses. Wait at least 15 minutes after using the eye drops before putting your lenses back in. There is a preservative in TOBRAVISC (benzododecinium bromide) that can affect soft contact lenses.
The usual dose
The usual dose in adults and children 1 year
and older is 1 drop in your eye(s), twice a day-morning and evening for 7 days
In severe infections, this may be ircreased to 1 drop 4 times a day for the first day, followed by 1 drop twice a day for 6 days.
Do not stop using TOBRAVISC before your doctor advises, even if your symptoms improve.
Remove the loose collar from the cap when the bottle is first opened.
Always use TOBRAVISC exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
How to use
• Wash your hands before you start.
• Twist off the bottle cap.
• Hold the bottle pointing down, between your thumb and fingers.
• Tilt your head back.
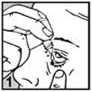
• Pull down your lower eyelid with a finger, until there is a ‘pocket’ between the eyelid and your eye. The drop will go in here (picture 1).
• Bring the bottle tip close to the eye. Do this in front of a mirror if it helps.
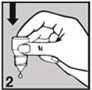
Do not touch your eye or eyelid.
surrounding areas or other surfaces with the dropper. It could infect the drops.
Gently press on the base of the bottle to release one drop at a time (picture 2).
Don’t squeeze the bottle, only a gentle press on the bottom is needed.
If you use drops in both eyes, repeat the steps for your other eye. Put the bottle cap firmly back on immediately after use.
• If a drop misses your eye, try again.
• If you forget to use TOBRAVISC, do not
worry, just use it as soon as possible.
However, if it is almost time for your next dose, skip the missed dose and go back to your normal dosing schedule. Do not take a double dose to make up.
• If you use more TOBRAVISC than you should, it can be. rinsed out with warm water.
If you have any further questions on the use
of TOBRAVISC. ask your doctor or pharmacist.
Like all medicines. TOBRAVISC can cause side effects, although not everybody gets them.
The following side effects are common and are experienced by up to 1 in 10 people using TOBRAVISC: eye redness, eye discomfort, eye allergy, itchy eyelids.
The following uncommon side effects have been reported by up to 1 in 100 people using TOBRAVISC:
• headache, hypersensitivity reactions
• inflammation, scratching or other problems with the surface of the eye such as swelling
• impaired or blurred vision
• redness or swelling of the eyelid
• eye discharge, eye pain, dry eyes, itchy eyes, watery eye(s)
• problems with the skin such as abnormal sensations, inflammation of the skin, itchy or dry skin
• the appearance of patches on the skin
• loss of eyebrows or eyelashes
The following side effects have also been reported but it is not known how many people have been affected:
• eye irritation
• severe allergic reactions
• serious skin reactions (Stevens-Johnson syndrome and erythema multiforme).
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
• Keep out of the reach and sight of children.
• Keep the bottle tightly closed.
• Do not use the drops after the expiry date (marked ‘Exp’) on the bottle and the carton. The expiry date refers to the last day of that month.
• Stop using the bottle 4 weeks after first opening, to prevent infections.
• Medicines should not be disposed of via waste water or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
• Do not pass this medicine on to others. It
may harm them, even if their symptoms are the same as yours.
What TOBRAVISC contains
• The active substance is tobramycin 3 mg/ml.
• The other ingredients are xanthan gum, benzododecinium bromide, mannitol, trometamol, boric acid, polysorbate 80, sulphuric acid and/or sodium hydroxide (to adjust pH) and purified water.
What TOBRAVISC looks like and contents of the pack
TOBRAVISC is a clear solution supplied in a pack containing a 5 ml plastic bottle with a screw cap.
Marketing authorisation holder:
Alcon Laboratories (UK) Ltd.
Frimley Business Park,
Frimley, Camberley, Surrey,
GU16 7SR, United Kingdom.
Manufacturer:
SA Alcon-Couvreur NV
Rijksweg 14, B-2870 Puurs, Belgium.
This leaflet was last revised in March 2016.
©2009, 2014.2016 Novartis 03-2016
45602-2