Ventolin Injection 500mcg/1ml
Out of date information, search anotherSUMMARY OF PRODUCT CHARACTERISTICS
1 NAME OF THE MEDICINAL PRODUCT
Ventolin Injection 500 micrograms (0.5mg) in 1ml
2. Qualitative and Quantitative Composition
Ventolin Injection 500 micrograms (0.5mg) in 1ml (500 micrograms/ml) is presented as ampoules of 1ml, each containing 500 micrograms salbutamol as salbutamol sulfate BP in a sterile isotonic solution.
3 PHARMACEUTICAL FORM
A colourless or faintly straw coloured solution for injection.
4 CLINICAL PARTICULARS
4.1 Therapeutic indications
Ventolin Injection is indicated in adults and adolescents.
Ventolin Injection provides short-acting (4-6 hour) bronchodilation with a fast onset (within 5 minutes) in reversible airways obstruction. It is indicated for the relief of severe bronchospasm.
4.2 Posology and method of administration
Ventolin Injection may be administered by the subcutaneous, intramuscular or intravenous route, under the direction of a physician.
Adults:
Subcutaneous route: 500 micrograms (8 micrograms/kg body weight) and repeated every four hours as required.
Intramuscular route: 500 micrograms (8 micrograms/kg body weight) and repeated every four hours as required.
Slow intravenous injection:
250 micrograms (4 micrograms/kg bodyweight) injected slowly. If necessary the dose may be repeated.
The use of Ventolin Injection 500 micrograms in 1ml (500 micrograms/ml, for intravenenous administration may be facilitated by dilution to 10ml with Water for Injection BP (final concentration of 50 micrograms/ml) and 5mls of the diluted preparation (250 micrograms/5ml) administered by slow intravenous injection.
Paediatric Population
The safety and efficacy of Ventolin Injection in children under the age of 12 has not been established. From the available data no recommendation on posology can be made.
Children aged 12 years and over: Dose as per adult population Instructions to open the ampoule
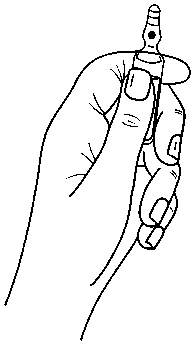
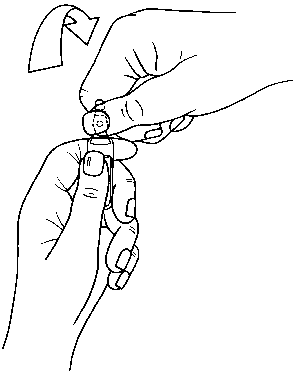
Ampoules are equipped with the OPC (One Point Cut) opening system and must be opened using the following instructions:
. hold with one hand the bottom part of the ampoule as indicated in Picture 1 . put the other hand on the top of the ampoule positioning the thumb above the coloured point and press as indicated in Picture 2
Picture 1
Picture 2
4.3. Contra-indications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1.
Non-i.v. formulations of salbutamol must not be used to arrest uncomplicated premature labour or threatened abortion.
4.4 Special warnings and precautions for use
Bronchodilators should not be the only or main treatment in patients with severe or unstable asthma. Severe asthma requires regular medical assessment, including lung-function testing, as patients are at risk of severe attacks and even death. Physicians should consider using the maximum recommended dose of inhaled corticosteroid and/or oral corticosteroid therapy in these patients.
The dosage or frequency of administration should only be increased on medical advice.
Patients being treated with Ventolin Injection may also be receiving shortacting inhaled bronchodilators to relieve symptoms. Increasing use of bronchodilators, in particular short-acting inhaled p2-agonists to relieve symptoms, indicates deterioration of asthma control.
The patient should be instructed to seek medical advice if short-acting relief bronchodilator treatment becomes less effective, or more inhalations than usual are required. In this situation the patient should be assessed and consideration given to the need for increased anti-inflammatory therapy (e.g. higher doses of inhaled corticosteroid or a course of oral corticosteroid).
Cardiovascular effects may be seen with sympathomimetic drugs, including salbutamol. There is some evidence from post-marketing data and published literature of rare occurrences of myocardial ischaemia associated with salbutamol. Patients with underlying severe heart disease (e.g. ischaemic heart disease, arrhythmia or severe heart failure) who are receiving salbutamol should be warned to seek medical advice if they experience chest pain or other symptoms of worsening heart disease. Attention should be paid to assessment of symptoms such as dyspnoea and chest pain, as they may be of either respiratory or cardiac origin.
Salbutamol should be administered cautiously to patients suffering from thyrotoxicosis.
Potentially serious hypokalaemia may result from P2-agonist therapy, mainly from parenteral and nebulised administration. Particular caution is advised in acute severe asthma as this effect may be potentiated by hypoxia and by concomitant treatment with xanthine derivatives, steroids and diuretics.
Serum potassium levels should be monitored in such situations.
Severe exacerbations of asthma must be treated in the normal way.
The use of Ventolin Injection in the treatment of severe bronchospasm does not obviate the requirement for corticosteroid therapy as appropriate. When practicable, administration of oxygen concurrently with Ventolin Injection is recommended. In common with other P-adrenoceptor agonists, salbutamol can induce reversible metabolic changes such as hypokalaemia and increased blood glucose levels. Diabetic patients may be unable to compensate for the increase in blood glucose and the development of ketoacidosis has been reported. Concurrent administration of corticosteroids can exaggerate this effect. Diabetic patients and those concurrently receiving corticosteroids should be monitored frequently.
Lactic acidosis has been reported in association with high therapeutic doses of intravenous and nebulised short-acting beta-agonist therapy, mainly in patients being treated for an acute asthma exacerbation (see Section 4.8). Increase in lactate levels may lead to dyspnoea and compensatory hyperventilation, which could be misinterpreted as a sign of asthma treatment failure and lead to inappropriate intensification of short-acting beta-agonist treatment. It is therefore recommended that patients are monitored for the development of elevated serum lactate and consequent metabolic acidosis in this setting.
As maternal pulmonary oedema has been reported during or following management of premature labour with P2-agonists, careful attention should be given to fluid balance and cardio-respiratory function should be monitored. If signs of pulmonary oedema develop, discontinuation of treatment should be considered (see section 4.8).
4.5. Interaction with other Medicinal Products and other Forms of Interaction
Salbutamol and non-selective beta-blocking drugs such as propranolol, should not usually be prescribed together.
4.6. Pregnancy and Lactation
Pregnancy
Administration of drugs during pregnancy should only be considered if the expected benefit to the mother is greater than any possible risk to the foetus.
As with the majority of drugs, there is little published evidence of the safety of salbutamol in the early stages of human pregnancy, but in animal studies there was evidence of some harmful effects on the foetus at very high dose levels.
Breast-feeding
As salbutamol is probably secreted in breast milk its use in nursing mothers is not recommended unless the expected benefits outweigh any potential risk. In such situations the use of the inhaled route may be preferable although it is not known whether salbutamol has a harmful effect on the neonate.
Fertility
There is no information on the effects of salbutamol on human fertility. There were no adverse effects on fertility in animals (see section 5.3).
4.7. Effects on Ability to Drive and Use Machines
Not applicable.
4.8. Undesirable Effects
Adverse events are listed below by system organ class and frequency. Frequencies are defined as: very common (>1/10), common (>1/100 and <1/10), uncommon (>1/1000 and <1/100), rare (>1/10,000 and <1/1000) and very rare (<1/10,000). Very common and common events were generally determined from clinical trial data. Rare, very rare and unknown events were generally determined from spontaneous data.
Immune system disorders
Very rare: Hypersensitivity reactions including angioedema, urticaria,
bronchospasm, hypotension and collapse.
Metabolism and nutrition disorders
Rare: Hypokalaemia.
Potentially serious hypokalaemia may result from beta-agonist therapy.
Nervous system disorders
Very common: Tremor.
Common: Headache.
Very rare: Hyperactivity.
Cardiac disorders
Very common:
Common:
Rare:
Unknown:
Tachycardia.
Palpitations.
Cardiac arrhythmias including atrial fibrillation, supraventricular tachycardia and extrasystoles.
Myocardial ischaemia* ( see section 4.4)
Vascular disorders
Rare: Peripheral vasodilatation.
Respiratory, thoracic and mediastinal disorders
Uncommon: Pulmonary oedema.
In the management of pre-term labour, Ventolin Injection has uncommonly been associated with pulmonary oedema. Patients with predisposing factors including multiple pregnancies, fluid overload, maternal infection and preeclampsia may have an increased risk of developing pulmonary oedema.
Gastrointestinal disorders
Unknown: Nausea, vomiting *.
Musculoskeletal and connective tissue disorders
Common: Muscle cramps.
Injury, poisoning and procedural complications
Unknown: Slight pain or stinging on intramuscular use of undiluted
injection*.
* reported spontaneously in post-marketing data therefore frequency regarded as
unknown.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard.
4.9. Overdose
The most common signs and symptoms of overdose with salbutamol are transient beta agonist pharmacologically mediated events, including tachycardia, tremor, hyperactivity and metabolic effects including hypokalaemia and lactic acidosis (see sections 4.4 and 4.8).
Hypokalaemia may occur following overdose with salbutamol. Serum potassium levels should be monitored.
Lactic acidosis has been reported in association with high therapeutic doses as well as overdoses of short-acting beta-agonist therapy, therefore monitoring for elevated serum lactate and consequent metabolic acidosis (particularly if there is persistence or worsening of tachypnea despite resolution of other signs of bronchospasm such as wheezing) may be indicated in the setting of overdose.
Nausea, vomiting and hyperglycaemia have been reported, predominantly in children and when salbutamol overdose has been taken via the oral route.
Further management should be as clinically indicated or as recommended by the national poisons centre, where available.
5.1. Pharmacodynamic Properties
Pharmacotherapeutic group: Selective beta-2-adrenoreceptor agonists ATC Code: R03CC02
Salbutamol is a selective beta-2 adrenoceptor agonist. At therapeutic doses it acts on beta-2 adrenoceptors of bronchial muscle providing short-acting (4-6 hour) bronchodilation in reversible airways obstruction.
5.2. Pharmacokinetic Properties
Salbutamol administered intravenously has a half-life of 4 to 6 hours and is cleared partly renally and partly by metabolism to the inactive 4’-O-sulfate (phenolic sulfate) which is also excreted primarily in the urine. The faeces are a minor route of excretion. The majority of a dose of salbutamol given intravenously, orally or by inhalation is excreted within 72 hours. Salbutamol is bound to plasma proteins to the extent of 10%.
5.3. Preclinical Safety Data
In common with other potent selective P2-agonists, salbutamol has been shown to be teratogenic in mice when given subcutaneously. In a reproductive study, 9.3% of fetuses were found to have cleft palate at 2.5mg/kg dose, 4 times the maximum human oral dose. In rats, treatment at the levels of 0.5, 2.32, 10.75 and 50mg/kg/day orally throughout pregnancy resulted in no significant fetal abnormalities. The only toxic effect was an increase in neonatal mortality at the highest dose level as the result of lack of maternal care. Reproductive studies in the rabbit at doses of 50mg/kg/day orally (i.e. much higher than the normal human dose) have shown fetuses with treatment related changes; these included open eyelids (ablepharia), secondary palate clefts (palatoschisis), changes in ossification of the frontal bones of the cranium (cranioschisis) and limb flexure.
In an oral fertility and general reproductive performance study in rats at doses of 2 and 50 mg/kg/day, with the exception of a reduction in number of weanlings surviving to day 21 post partum at 50 mg/kg/day, there were no adverse effects on fertility, embryofetal development, litter size, birth weight or growth rate.
6.1. List of Excipients
Sodium chloride Sodium hydroxide pellets Dilute sulfuric acid Water for injections Nitrogen (oxygen free)
6.2. Incompatibilities
None stated
6.3. Shelf Life
36 months
24 hours - shelf life of admixtures with infusion fluids.
6.4.
Special Precautions for Storage
Store below 30°C and keep the ampoule in the outer container in order to protect from light.
6.5. Nature and Contents of Container
Clear, neutral glass ampoules, packed in plastic trays with a cardboard sleeve over the trays.
Pack size: 1 ml ampoules in plastic trays of 5.
6.6. Special precautions for disposal and other handling
The only recommended diluents for Ventolin Injection are water for injections BP, sodium chloride injection BP, sodium chloride and dextrose injection BP or dextrose injection BP.
All unused admixtures of Ventolin Injection should be discarded 24 hours after preparation.
Ventolin Injection should not be administered in the same syringe as any other medication.
7 MARKETING AUTHORISATION HOLDER
Glaxo Wellcome UK Ltd trading as GlaxoSmithKline UK Stockley Park West Uxbridge
Middlesex UB11 1BT.
8. MARKETING AUTHORISATION NUMBER(S)
PL 10949/0084
9. DATE OF FIRST AUTHORISATION / RENEWAL OF THE AUTHORISATION
8 November 1993/20 October 1995
10 DATE OF REVISION OF THE TEXT
02/03/2015